Clinical trials for chronic wounds have long struggled to efficiently collect consistent wound metrics and compare subjective assessments that can vary across clinicians and sites. The Wound Care Collaborative Community (WCCC) is advocating for a modernized approach. They are seeking a future of skin and wound clinical trials that embraces objective tools and richer data. Swift is fully aligned with this critical transformation.
The Need for Consistent, Objective Wound Data
The right tools need to be used to reduce subjectivity across clinicians and to improve wound clinical trial data reliability. This includes images calibrated for lighting, distance and color as well as AI-powered features that provide greater consistency (intra-reliability) than traditional assessments between clinicians. Without utilizing powerful, validated technology, research data is prone to significant errors and inconsistencies, ultimately slowing down drug and device development.
Advanced Imaging for Deeper Insights
Not every part of wound healing is visible to the naked eye. New technologies, like thermal and bacterial imaging, may serve as early indicators of wound progression or complication.1,2,3,4 This is especially important when chronic wound closure can take months or even years. These deeper insights and data points offer a chance to provide more robust evidence of treatment effects.
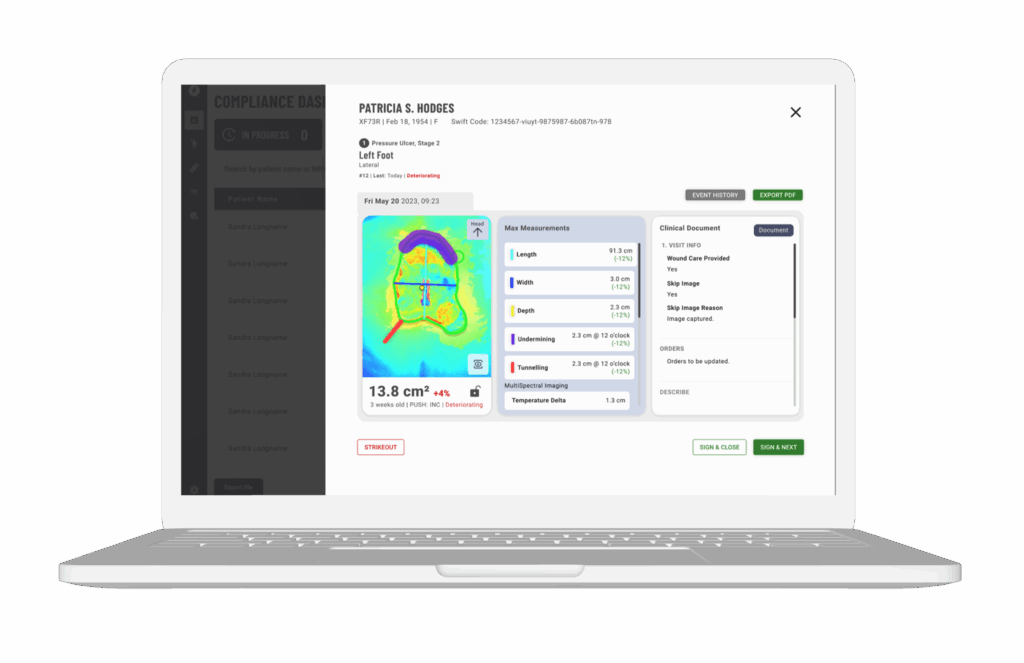
Expanded Data Beyond Simply Closure
Wound closure and area reduction are the most common data points in wound care clinical trials, but they don’t tell the whole story. Patient outcomes such as infection reduction, pain relief, improved mobility and reduced rates of recurrence or amputation are proposed as key indicators of therapeutic success.5 Collecting data on these expanded metrics provides a more complete, holistic picture of a therapeutic’s true value.
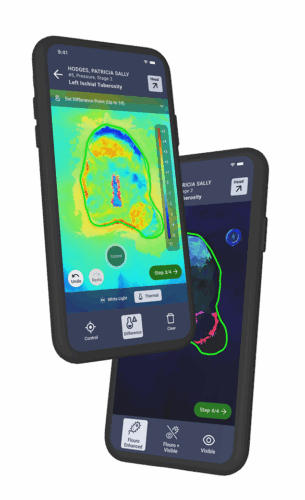
How Swift Helps Modernize Wound Research
Swift addresses the core challenges of wound care research, offering a platform that delivers objective, comprehensive, scalable and advanced data collection. We can help skin and wound clinical trial teams to:
- Capture standardized wound images and objective data to ensure consistency.
- Calculate wound progression automatically and consistently.
- Enable expanded data collection in a standardized manner.
- Provide real-time analytics for compliance monitoring.
- Gain insight and oversight into clinical data collection by study sponsors or CROs.
- Add advanced imaging insights, great for case studies.
Learn how Swift Ray® 1 can support your next skin or wound clinical trial.
- Annadatha, A., G. Danagoulian, J. Muser, et al. “Infrared Thermography in Dermatology: Current Applications and Future Directions.” Journal of the European Academy of Dermatology and Venereology, vol. 36, no. 1, 2022, pp. 13–24
- Cai, F., X. Jiang, X. Hou, et al. “Application of Infrared Thermography in the Early Warning of Pressure Injury: A Prospective Observational Study.” Journal of Clinical Nursing, vol. 30, no. 3–4, 2021, pp. 559–567.
- Lopez, A. J., Jones, L.M., Reynolds, L. Diaz, R. C., George, I. K., Little, W., Fleming, D., D’souza, A., Rennue, M. Y., Rumbaugh, K. P., Smith, A.C.. (2021). Detection of bacterial fluorescence from in vivo wound biofilms using a point-of-care fluorescence imaging device. International Wound Journal, 18(5), 626–638. https://doi.org/10.1111/iwj.13564
- Serena, T. E., Snyder, R. J., Bowler, P. G. (2023). Fluorescence-informed biopsy improves microbial profiling: Sensitivity 98.7% vs. clinical judgment 87.2% in detecting >104 CFU/g. Frontiers in Cellular and Infection Microbiology, 12, 1070311. https://doi.org/10.3389/fcimb.2022.1070311
- Cole W, Sanders M, Driver V, et al. Wound Care Collaborative Community Recommendations for Chronic Wounds: FDA Guidance Update on Clinical Trial Design. Presented at the Symposium on Advanced Wound Care Fall 2025, Las Vegas, NV.





